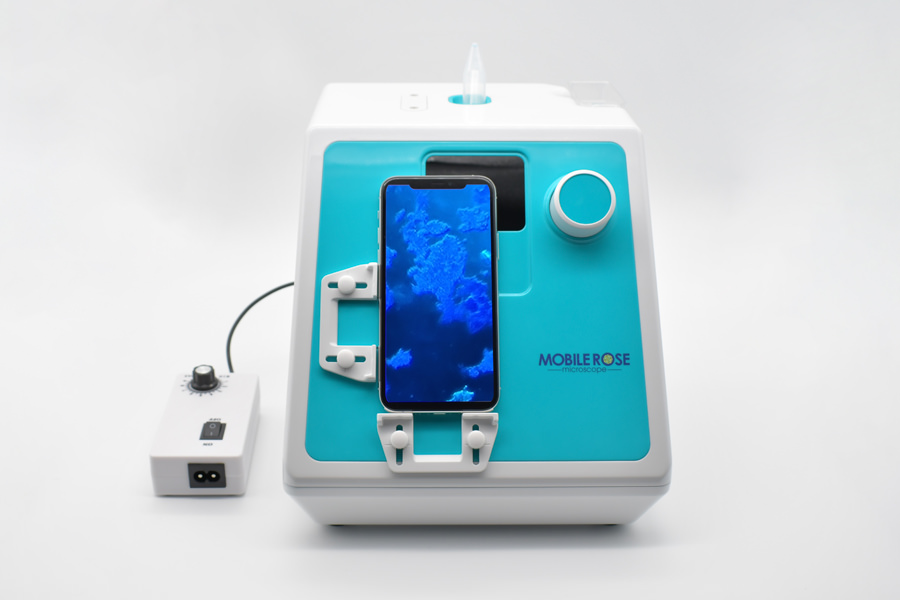
A joint paper with Kuma Hospital, one of the leading thyroid hospitals in Japan, was published in Endocrine Journal, a publication of the Japanese Endocrine Society.
According to this paper, 57.1% of the cases that were found to be inappropriate or unknown in the rapid adequacy assessment of thyroid gland by naked eyes were confirmed to have cells by using Mobile Rose and were considered appropriate.
The clinical utility of Mobile Rose has been shown to prevent unnecessary re-sampling.
For more information, please click on the link below.